You’re not the only one if you’re sick of living with chronic back pain. In fact, 31 million Americans suffer from back pain at some point, as stated by the American Chiropractic Association.
While there are many potential causes of back pain, one of the most frequent is a herniated disc. A herniated disc takes place when the gel-like center of a spinal disc rupture and puts pressure on the spinal nerve root. So, here are some quick tips to help alleviate the pain.
Sleep Better
The best sleeping position for alleviating back pain is typically considered to be sleeping on your side with your knees slightly bent. If it’s tough for you to sleep in this position, though, try lying on your back with a pillow under your knees. This will help maintain your back in a neutral position and take the pressure off your spine.
If you suffer from chronic back pain, you may also find it difficult to get enough sleep. This can create a vicious cycle, as poor sleep can worsen back pain and make it more difficult to get the rest you need. Sleeping on a mattress that is comfortably firm will help reduce this problem and provide additional support for your back.
Good Posture
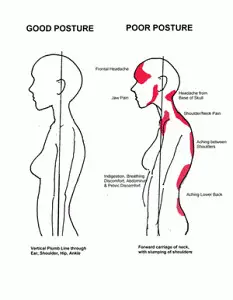 One of the easiest things you can do to reduce your back pain is to check your posture – how you stand, sit and move.
One of the easiest things you can do to reduce your back pain is to check your posture – how you stand, sit and move.
“Unconscious habits developed over the years are usually the cause of poor posture,” Nadine R. Ehler, MD, who is an associate professor of rehabilitation medicine at NYU Langone Health in New York City, states: “When we hunch and slouch our shoulders forward while sitting or standing for long periods of time, our head moves out of alignment with our center of gravity and puts tension on the muscles and ligaments in our necks and upper backs.”
Dr. Ehler explains that “good posture is like a reset button for your body.” When you stand up with proper posture, everything else falls into place. This includes your shoulders dropping to their natural position, your head coming off your neck, and your pelvis tilting forward so your lower back curves naturally. From there, everything else related to how you carry yourself, such as when you walk or sit at a desk, will also be corrected.
At first, it may not feel natural to be aware of all these different elements while you’re used to slouching forward (we know grandma wasn’t kidding about “standing up straight!”, but with a bit of practice, it will become second nature in no time.
Medication From the Store
You can purchase various kinds of medication from the store to assist with your back pain. These include nonsteroidal anti-inflammatory drugs (NSAIDs) to diminish inflammation and pain; counter-irritant creams, which work by causing a cooling or heating sensation that takes your mind off the pain; and topical pain relievers with ingredients like menthol or capsaicin to relieve pain.
NSAIDs can be bought without a prescription, but they might cause issues like indigestion, kidney problems, and bleeding. Topical pain relievers are usually safe to use, but they may not work as well as other treatments. If your back pain is bad or doesn’t go away with other treatments, you might need to see a doctor for stronger medication.
Prescription Pain Relievers
You may turn to a pain reliever if you’re experiencing back pain. Over-the-counter (OTC) medicines are drugs available without a doctor’s prescription. There are 3 types of OTC medicines that treat back pain and other painful muscle aches:
- Acetaminophen
- Nonsteroidal anti-inflammatory drugs (NSAIDs)
- Topical creams or ointments
Acetaminophen is often the first medication people turn to because it has fewer side effects than NSAIDs. If you’re thinking about taking NSAIDs, have a conversation with your doctor about the risks and benefits, as well as the correct dosage and duration of time you should take them. NSAIDs come in pill form (such as ibuprofen or naproxen) or topical form (for example, creams or gels that you rub into your skin).
There are additional medications that can assist with back pain, such as muscle relaxants and prescription painkillers. In more severe cases, your doctor may prescribe injections to help ease the pain. If other methods of treatment haven’t been effective, your doctor may recommend surgery.
Antidepressant Medications
Antidepressant drugs are sometimes used to manage chronic low back pain. They work by interfering with the chemical messengers that send pain signals from the nerves to the brain. By changing how these chemical messengers function, antidepressants can help lessen the experience of pain.
There are different types of antidepressant medications available, and they are grouped together based on their chemical structure. Some of the more commonly used types of antidepressants include tricyclic antidepressants, selective serotonin reuptake inhibitors (SSRIs), and monoamine oxidase inhibitors (MAOIs).
Tricyclic antidepressants are older drugs that have been used to treat a variety of conditions, including chronic pain, for many years. These drugs work by inhibiting the reuptake of both norepinephrine and serotonin, two important chemical messengers involved in transmitting pain signals. Amitriptyline (Elavil), imipramine (Tofranil), and nortriptyline (Pamelor) are all examples of tricyclic antidepressants.
Selective serotonin reuptake inhibitors (SSRIs) are a newer class of antidepressants that specifically target serotonin, another important chemical messenger involved in transmitting pain signals. SSRIs work by inhibiting the reuptake of serotonin, which increases its availability in the brain. This action is thought to reduce pain perception. Examples of SSRIs include fluoxetine (Prozac), paroxetine (Paxil), and sertraline (Zoloft).
Monoamine oxidase inhibitors (MAOIs) are another type of antidepressant that focuses on monoamine neurotransmitters, such as norepinephrine and serotonin. These drugs function by inhibiting the enzyme monoamine oxidase, which breaks down these neurotransmitters. This action raises the levels of norepinephrine and serotonin in the brain, which is believed to lower pain perception. Some MAOIs are phenelzine (Nardil) and tranylcypromine (Parnate).
Physical Therapy
If you want to find relief from back pain, physical therapy may be a good option. Physical therapists have extensive knowledge about proper alignment, movement, and posture. They can instruct you on the best way to sit, stand, lift and sleep to maintain your spine in the correct alignment and help prevent more back pain.

Physical therapists can also help you strengthen your core muscles, which support your spine. A strong core helps prevent back pain by keeping your spine properly aligned. Physical therapists can also use massage, heat or cold therapy, and other techniques to relieve back pain.
Don’t Rest an Achy Back
Though you may want to simply stay in bed and rest when you’re experiencing back pain, that’s actually one of the worst things you can do for your back.
“The best way to treat acute low back pain is to keep moving,” says Erin Michos, M.D., M.H.S., an assistant professor of medicine at Johns Hopkins University School of Medicine and a spine specialist at Johns Hopkins Bayview Medical Center. “We tell patients, ‘If you can stand and walk, do it.'”
This advice should be followed for good reason. ” Bed rest can cause weight gain, increase your risk for blood clots, lead to joint stiffness and deconditioning, interrupt your sleep, and make you cranky,” Dr. Michos says. “It also prolongs recovery.”
Studies have actually demonstrated that people who don’t take it easy and instead stay active have less pain and recover more rapidly than those who stay in bed. They also have fewer other issues, such as depression.
Ice and Heat
The two most common ways to treat back pain at home are with ice and heat. You can switch between the two methods, or use whichever one seems to work better for you at the moment.
Applying ice to the affected area can help reduce swelling and pain. Put an ice pack (or a bag of frozen peas wrapped in a thin towel) on the painful area for 15-20 minutes, several times a day.
Heat: Applying heat to the affected area can help muscles relax and ease the pain. Use a heating pad on the lowest setting, or take warm baths several times a day. Apply heat to the affected area for 15-20 minutes at a time.
If your back pain does not improve or gets worse after a few days of self-care, call your doctor.
Hands-On Therapy
A recent study found that hands-on therapy may provide temporary relief from back pain caused by structural issues. In the study, people with back pain who received weekly massages for a 10-week period reported less pain and disability than those who didn’t receive massage therapy.
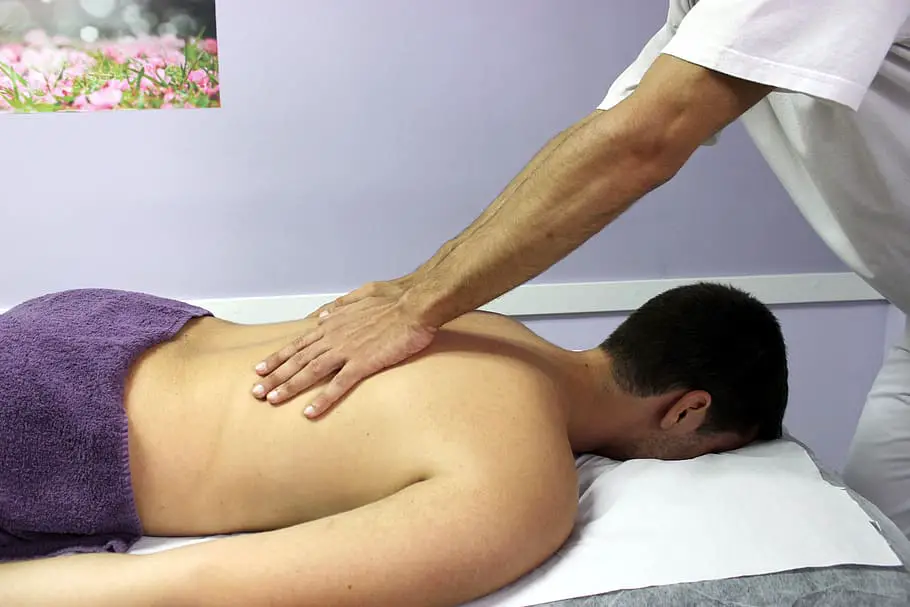
If you’re thinking about massage therapy to ease back pain, talk to your doctor first. After that, look for a licensed specialist who has experience treating people with back issues. You might need multiple massage sessions to get relief.
Nerve Stimulation
Nerve stimulation is a potential pain treatment that your doctor could suggest as part of your plan. Nerve stimulation involves sending electrical pulses to the nerves to stop them from sending pain signals to the brain. There are two types of nerve stimulation commonly used to treat pain: transcutaneous electrical nerve stimulation (TENS) and percutaneous electrical nerve stimulation (PENS).
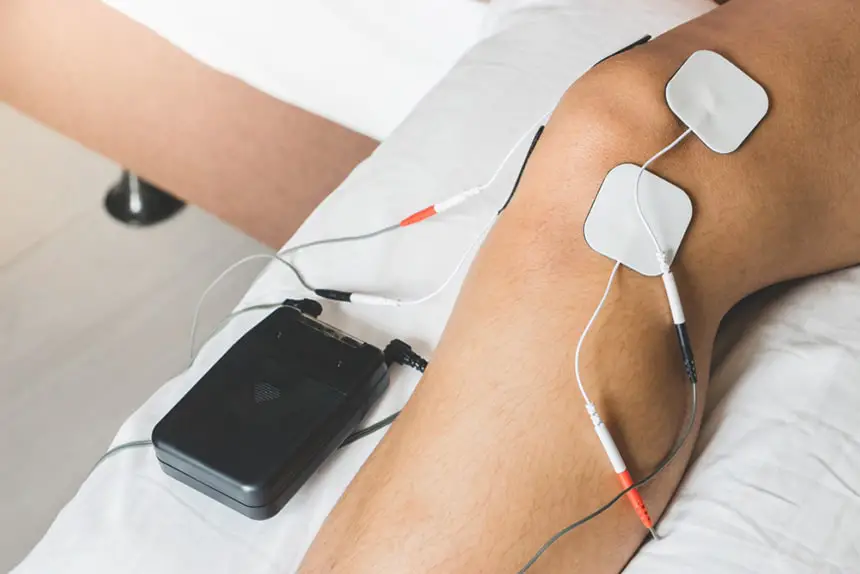
TENS is a technique that employs a small, handheld device to deliver electrical pulses through wires that are connected to sticky pads placed on the skin. PENS is comparable, but the electrical pulses are transmitted through a needle that is inserted into the skin. TENS and PENS are typically outpatient procedures, which means you can go home on the same day of the treatment.
Depending on the type of pain you’re experiencing, your doctor may suggest particular treatments. For instance, if you’re struggling with back pain, TENS or PENS might be part of your conservative care plan. Conservative care is a nonsurgical method of treating pain that encompasses physical therapy, exercises, and medications.
If you have chronic pain, your doctor might also recommend other types of nerve stimulation, such as dorsal root ganglion stimulation (DRGS) or spinal cord stimulation (SCS). DRGS is a newer type of nerve stimulator that sends electric pulses directly to the dorsal root ganglion, which is a cluster of nerves in the spine. SCS implantable devices deliver electric pulses to the spinal cord to help block incoming pain signals from reaching the brain.
What Is Inversion Therapy?
Inversion therapy is a unique form of physical therapy that involves hanging upside down or at an inverted angle. This therapy has been used for centuries to treat a variety of health conditions, including back pain, joint pain, and headaches.
Inversion therapy can be performed using a variety of methods, including inversion tables, gravity boots, and hanging upside down from a bar. The key to inversion therapy is to hang upside down for a short period of time, usually no more than two minutes at a time. The goal is to use the effects of gravity to decompress the spine and relieve pressure on the vertebrae, discs, and nerves.
There is some evidence to suggest that inversion therapy may be effective for treating back pain. One study found that inversion therapy was able to reduce pressure on the spine by up to 50 percent. Additionally, inversion therapy has been shown to help with sciatica, improve blood circulation and reduce inflammation.
If you are interested in trying inversion therapy, it is important to consult with your doctor first. Inversion therapy may not be suitable for people with certain medical conditions, such as high blood pressure or glaucoma. Additionally, it is important to use inversion therapy devices carefully and follow manufacturers’ instructions to avoid injuries.
How does inversion therapy work?
Inversion therapy is a type of physical therapy that involves hanging upside down or being in a position where your head is lower than your heart. This technique is sometimes used to treat back pain, neck pain, and headaches.
The theory behind inversion therapy is that it can help relieve pressure on your spine and other structures in your back. This type of therapy can also help increase blood flow to your head and reduce pressure on your blood vessels.
Inversion therapy is not for everyone, and it’s important to talk to your doctor before you try this type of therapy. In some cases, inversion therapy can cause serious injuries, so it’s important to make sure you are healthy enough for this type of activity.
What are the benefits of inversion therapy?
Inversion therapy is a form of treatment that involves hanging upside down or at an inverted angle.
The practice is said to offer a range of benefits, such as:
- Reducing back pain
- Relieving pressure on the spine
- Improving circulation
- Increasing flexibility
- Easing tension headaches
A number of studies have shown that inversion therapy can be effective in relieving back pain. However, more research is needed to confirm these findings.
How do I know if inversion therapy is right for me?
There are a few things you should consider before trying inversion therapy. First, inversion therapy is not for everyone. If you have any of the following conditions, you should not try inversion therapy:
- High blood pressure
- Heart disease
- Glaucoma
- History of stroke
- Herniated disc or other back problems
- Surgery on the spine or back within the past 6 months
In addition, if you are pregnant, you should not try inversion therapy. If you have any other health concerns, be sure to talk to your doctor before trying inversion therapy.
Assuming you don’t have any of the above conditions, inversion therapy can be a great way to relieve pain and improve your overall health.
How can I get started with inversion therapy?
If you’re interested in inversion therapy, there are a few things you can do to get started.
First, talk to your doctor. Inversion therapy may not be right for you if you have certain medical conditions, such as high blood pressure, glaucoma, or a history of stroke.
Second, try an inversion table. Inversion tables are devices that allow you to hang upside down, and they’re widely available for purchase online and in stores.
Third, start slowly. If you’re new to inversion therapy, it’s important to take things slowly at first. Start with brief sessions (no more than three minutes at a time) and gradually increase the amount of time you spend inverted as your body adjusts.
Finally, be sure to use proper form. When inverting, be sure to keep your back straight and your chin tucked into your chest. Avoid jerking or swinging motions, and never invert further than is comfortable for you.
NEXT: Read our article about the 3 best inversion tables of [2022].
Scoliosis Diagnosis and Treatment
Scoliosis is a sideways curvature of the spine. However, there’s no need to fret – there are treatments available to correct it.
Diagnosis
Parents or guardians are often the first to detect scoliosis when they notice that the child’s shoulders or hips are not even. The child may also stand with one hip higher than the other, or the ribs may be more visible on one side of the back. If you observe any of these signs, take your child to see a doctor.
The doctor will ask about the child’s medical history and any symptoms they may have, such as numbness, tingling, or pain in the back or extremities. They will also do a physical examination, during which they will ask the child to stand and then bend forward at the waist. The doctor will look for any asymmetry in the spine and check for abnormal reflexes.
If scoliosis is a possibility, your doctor may send you to a pediatric orthopedist or neurologist for additional testing. This could involve a neurological exam to look for muscle weakness or numbness and X-rays to assess the spine. If scoliosis is diagnosed, treatment will depend on how severe the curve is and if it is still growing.
Imaging Tests
Although imaging tests are vital for diagnosing scoliosis, they aren’t perfect. The physical examination provides a more accurate measure of the curve than imaging tests. Also, you will be exposed to some radiation if you get an imaging test.
X-rays are the most typical imaging test for scoliosis. They can demonstrate the extent of the curvature and if the curve is worsening. A single x-ray can only show a limited part of the spine, so your doctor may request multiple x-rays to get a better idea.
Although plain x-rays have certain drawbacks, such as being difficult to interpret (particularly in growing children) and not providing a three-dimensional image of the spine like other tests, they have one significant advantage: you are exposed to very little radiation.
Another test that can display the three-dimensional shape of the spine and any atypical spinal cord or nerve roots is MRI (magnetic resonance imaging). However, MRI employs powerful magnets and doesn’t give off radiation, making it a better option for children and pregnant women. Additionally, MRI is quite costly so it isn’t always used unless there’s reason to believe there’s something else amiss in addition to scoliosis.
Your doctor may order an ultrasound if they believe you have spinal cord compression from your scoliosis curve. Ultrasound uses sound waves to create images, so it is a radiation-free exam. This test is often done in conjunction with an MRI or CT scan (computerized tomography).
Treatment
The three most frequent treatments for scoliosis are observation, surgery, and bracing. The recommended treatment for a particular patient is based on the intensity of the curve, the patient’s age, and gender, and if the curve is worsening.
Mild scoliosis, which is a spinal curvature of 10 degrees or less, can often be treated with regular checkups. The doctor will measure the curve during these visits to see if it has gotten worse. If the curve is not progressing and the patient is still growing, observation is usually continued. In some cases, though, the doctor may recommend bracing for patients with very mild scoliosis, especially if there is a family history of larger curves.
For patients with curves between 25 and 40 degrees, bracing is typically recommended. The objective of bracing is to halt the progression of the curve and stop it from worsening. It is most successful in patients who are still growing and whose bones have not yet reached full maturity.
There are a few different types of braces that can be used to treat scoliosis. The type of brace prescribed will depend on the severity of the curve and other factors such as the patient’s age, sex, and lifestyle.
If a patient has a curve greater than 40 degrees, or if the curve is progressing despite treatment with a brace, surgery is usually recommended. This involves correcting the spinal curvature by fusing some of the bones in the spine together (spinal fusion).
The goal of surgery is to stop the progression of the curve and to prevent further deformity. Surgery can be very effective in treating scoliosis; however, it does have some risks and potential complications that should be discussed with your doctor prior to making a decision about treatment.
Braces
The majority of braces are constructed from high-tech plastics that are practically unnoticeable under clothing. They are also lightweight and comfortable, so people usually wear them all day and night, taking them out only for bathing or physical activities. Although there are a few restrictions to wearing a brace, the majority of people can still participate in most activities.

The most typical treatment for scoliosis is braces. They are generally recommended for kids who have stopped growing (usually girls aged 10-14 and boys aged 11-15). The usefulness of braces improves as the child gets closer to finishing growth. For moderate scoliosis, the objective of treatment is to prevent the curve from worsening. In some instances, the curve improves while the child is wearing the brace.
Depending on the patient’s needs, there are various types of braces. The two most frequent types are Boston braces and Chesapeake braces. Those with curves in their upper spine require Boston braces, while Chesapeake braces are for patients with curves in either their lower spine or upper spine.
It is still important to participate in physical activities, like sports, even when wearing a brace. Exercises that target and strengthen back and abdominal muscles may also be recommended.
Surgery
Spinal surgery to correct scoliosis is a very serious matter and should only be considered after all other treatment options have failed. This type of surgery is most often performed on patients who have severe scoliosis that is still progressing and who are still growing.
Surgery’s objective is to halt the curve’s progression and improve the spine’s appearance and function. Surgery is not usually done to achieve perfect alignment, but rather to keep the curve from worsening.
Surgery for this condition involves placing metal rods next to the spine, which is then secured in place. The rods can be either straight or curved and are made out of titanium or stainless steel. They are attached to the spine with screws, hooks, or wires.
The most typical surgical operation for scoliosis is referred to as posterior instrumented fusion (PIF). This involves putting metal rods on the back of the spine through tiny incisions. The rods are then secured to the vertebrae with screws or wires. A bone graft might also be used to fuse together the vertebrae above and below the curve.
AIF, or anterior instrumented fusion, is another type of scoliosis surgery, but it isn’t performed as often as PIF. In this operation, metal rods are placed on the front side of the spine through tiny incisions. The rods are secured to the vertebrae with screws or wires, and a bone graft may also be employed.
Expandable rods for scoliosis surgery are a newer option that has some advantages over traditional metal rods. With expandable rods, surgeons can fix a bigger curve during one operation, and they can be lengthened if necessary over time.
These rods are also less likely to require revision surgery in the future. Expandable rods are made of titanium or cobalt chrome, and they are typically covered with a layer of polyethylene (plastic). Expandable rods come in different sizes and shapes to fit each patient’s needs.
Remote-controlled expandable rods are a newer type of expandable rod that can be lengthened or shortened after surgery using a remote control outside the body. This allows for more precise control over rod length and eliminates the need for revision surgery in most cases.
Remote-controlled expandable rods are made of titanium or cobalt chrome, and they are usually covered with a layer of polyethylene (plastic). These rods come in different sizes and shapes to fit each patient’s needs.
There are certain risks that come along with scoliosis surgery, such as infection, blood loss, nerve damage, and complications from anesthesia. There is also a possibility that the curve may worsen after surgery in spite of correction with metal implants or other devices such as Harrington Rods™️.
If you are thinking about scoliosis surgery, it is essential to talk about all potential risks and benefits with your doctor before making a decision.
Lifestyle and Home Remedies
While there is no cure for scoliosis, treatment can often impede the condition from worsening. The kind of treatment will depend on the person’s age and how severe the curvature is.
For children with mild scoliosis, doctors typically suggest doing specific exercises to fortify the back and enhance posture. This care is called conservative management or nonsurgical treatment.
If the curve is more severe, your child might need to wear a back brace. A back brace is a garment that fits around the torso and helps hold the spine in a straighter position. It is usually made of tough plastic and has straps that go over the shoulders. A back brace must be worn all day, but can be removed for bathing and exercise.
The type of treatment for adults with scoliosis varies depending on the intensity of the condition and if it causes pain. If there’s no pain, possible treatments might only involve monitoring the condition through regular checkups. If the pain does develop, potential treatments could include physical therapy, medication prescribed by a doctor, or surgery.
Surgery is generally only recommended for scoliosis that is severe enough to cause pain or difficulty breathing. The objective of surgery is to prevent the curve from worsening and to alleviate pain. Surgery entails placing metal rods along the spine to keep it straight while it heals.
Alternative Medicine
There is presently no cure for scoliosis, but there are various treatment methods that can help manage the condition’s progression and ease symptoms. While traditional scoliosis treatments like wearing a back brace or having surgery may be necessary for more serious cases, there are also several alternative scoliosis treatments that may work well.
Some of the most frequently used alternative scoliosis treatments are chiropractic manipulation, electrical stimulation, and dietary supplements. By aligning the spine, chiropractic manipulation can take pressure off of the spinal cord. Electrical stimulation can help reduce muscle tension and pain. Dietary supplements can give the body vital nutrients that may be scarce because of scoliosis.
Before you decide on any kind of alternative scoliosis treatment, have a conversation with your doctor. Some alternative scoliosis treatments might not be right for all patients, and some might interfere with traditional scoliosis treatments such as back braces or surgery.
Preparing for Your Appointment
In order to get ready for your appointment, begin by making a list of every reason why you think your kid might need to see a physician. For example, has your child been injured or in pain recently?
After that, create a list of all the medications, vitamins, and supplements your child takes. This will assist the doctor in figuring out if there are any possible interactions between the medications.
You should also bring copies of any x-rays, MRIs, or other test results that have already been completed. In some cases, the doctor may need to order additional tests.
Make sure to ask how long the appointment will take and if there is anything you need to do to get ready when you book it. For instance, a lot of schools necessitate students to have a physical examination before they are able to compete in sports.
When you arrive for the appointment, be prepared to answer questions about your child’s medical history and current symptoms. The doctor will also likely perform a physical examination and order tests as necessary.
What You Can Do
There are a few things that can be done while waiting for an appointment with a doctor who specializes in treating scoliosis. First, if the curve is greater than 20 degrees, it is important to obtain a brace. The type of brace will be determined by the curvature of the spine and the age/growth potential of the child.
The goal of bracing is to prevent the curve from getting worse. It is important to note that braces do not correct existing curves, but rather they prevent progression.
The second thing to do is maintain good posture and alignment. This means standing up straight and keeping the shoulders level. Sitting up straight in chairs with both feet flat on the floor should become a habit. When lying down, it is best to sleep on one’s back with a pillow under the knees to maintain alignment of the spine.
What To Expect From Your Doctor
The doctor will probably ask you several questions when you visit them for your back pain.
The doctor will ask about the duration of your pain, and what makes it worse if you have any trouble breathing or feel any tingling or numbness in your extremities.
Your doctor will probably want to do a physical examination after taking your medical history. This will help them figure out if your pain is from muscle strain or something more severe, like scoliosis.
Your doctor will probably order an X-ray if they think you might have scoliosis. This will give them a clear picture of your spine and let them know if there is any curvature present.
Scoliosis is most often diagnosed in kids between the ages of 10 and 16. If it’s caught early on, treatments such as physiotherapy or braces can be successful in slowing down or halting the progression of the curve. In more serious cases, surgery might be required to fix the deformity.
What is Sciatica and How To Treat It?
Sciatica is a severe form of pain that starts in the lower back and extends down the leg, sometimes all the way to the foot. It occurs when there is pressure on the sciatic nerve, which is the longest nerve in your body. This pressure can be caused by a bone spur, a herniated disk, or pregnancy.
Sciatica can make it difficult to perform common activities such as sitting, standing, or walking. The silver lining is that the majority of people improve over time and don’t require surgery. Additionally, there are some helpful treatments that can ease the pain.
Sciatica can cause a sharp pain that gets worse when you sit or stand. You might also experience weakness, tingling, or numbness in your leg or foot. The pain might get worse when you cough or sneeze.
If you experience any of these symptoms, consult your doctor. They will be able to determine if you have sciatica and help you find the most effective treatment method.
Nonsurgical Options
The majority of people who have sciatica recover from the condition within a few weeks by using simple solutions such as over-the-counter pain medication and ice packs. However, some people may experience severe, chronic pain. If at-home remedies don’t alleviate your sciatica pain, your doctor may recommend nonsurgical sciatica treatments.
Some of the most frequent nonsurgical sciatica treatments are:
Physical therapy may be able to help relieve the pressure on your sciatic nerve and ease your pain. A physical therapist can teach you stretches and exercises to help relieve the pressure on your sciatic nerve. He or she may also use massage, electrical stimulation, or other techniques to ease your pain.
Occupational therapy may be able to help you find different ways to do your daily activities without aggravating your sciatica pain. For example, an occupational therapist may teach you how to sit in a chair or get in and out of bed without pain.
Injections. Your doctor may give you a steroid injection to help reduce the inflammation causing your sciatic nerve pain. In some cases, he or she may also inject antispasmodic medication into the muscles around your sciatic nerve to try to break the cycle of muscle spasms that often accompanies sciatica pain.
Physical Therapy
Sciatica is a condition that can result in discomfort in the lower back and legs. The Sciatic nerve is the longest nerve in the human body. It runs from the lower back, through the buttocks, and down the leg. Sciatica develops when this nerve becomes irritated or compressed.
Sciatica sufferers are often advised to seek physical therapy. A physical therapist has the ability to not only reduce pain but also improve the range of motion and bolster the muscles that hold up the spine. They may also suggest exercises to help stretch and fortify the muscles surrounding the sciatic nerve.
Stretching
Sciatic nerve pain relief can be achieved by stretching. Not only does stretching lengthen muscles, but it also raises muscle temperature, improves blood circulation, and prevents muscle tightness.
For a hamstring stretch, lie on your back with both legs extended and straight in front of you. Take one leg and bend it so the foot is flat on the floor next to the thigh of the other leg. With your back remaining flat on the floor, raise your other leg until it is pointing directly up at the ceiling.
You should feel a stretch in the back of your thigh and possibly in your lower back. Hold this position for 30 seconds before slowly lowering your leg back down to the starting position. Repeat with your other leg.
Exercise
If you have sciatica, it’s important to keep your body moving. Exercise may help to decrease inflammation and pain. A physical therapist can help you with form and make sure you’re doing the exercises correctly. Short walks are a good idea. Start with 10-15 minutes and gradually increase the time.
Limited Bed Rest
If you’re suffering from acute sciatica, you might find relief from taking short bed rest. You shouldn’t stay in bed for more than one or two days. This will help ease your pain and reduce the inflammation around the nerve. Once your pain is more manageable, you can start to slowly bring normal activities back into your life.
Additionally, steer clear of any physical activity that puts extra strain on your back, such as lifting heavy objects or running. If possible, sleep on a firm mattress to take some pressure off your lower back.
Hot and Cold Packs
If you’re in pain from sciatica, hot and cold packs may give you some relief. For the first few days, try applying cold packs to your lower back a few times each day for several minutes at a time. You can use ice packs, or put bags of frozen peas or corn in a towel and apply them to your back. After the first few days, switch to heat packs. You can use heating pads that you plug in, or put a hot water bottle on your back. Apply heat for 15 to 20 minutes at a time.
Alternative Therapies
Though there is no scientific evidence to back up claims that alternative therapies such as massage, chiropractic care, or acupuncture are effective for relieving sciatic nerve pain, many people say that these therapies help reduce pain and improve function.
Medicines
Sciatic nerve pain can be treated with various medications. The most typical and initial choice is over-the-counter pain medication, like acetaminophen or non-steroidal anti-inflammatory drugs (NSAIDs). These can provide limited relief but shouldn’t be taken for long periods of time. Other potential treatments include seizure medications, muscle relaxants, and steroid injections.
Sciatic nerve pain is often alleviated by steroid injections. They work to reduce the inflammation and swelling around the irritated nerve. Injections are most commonly administered every three to four weeks and are most successful when used in addition to other treatments, like physical therapy.
Elavil, a tricyclic antidepressant, is sometimes used to manage sciatic nerve pain. By blocking the reuptake of particular neurotransmitters, these drugs can help reduce pain. Some potential side effects are dry mouth, constipation, weight gain, and dizziness.
There are other potential countermeasures that can be used to alleviate sciatic nerve pain. These include acupuncture, massage therapy, yoga, and chiropractic care.
Surgery
Surgery for sciatica should only be considered after all other options have failed. In the majority of cases, conservative treatments will work. However, there are some instances where surgery might be the only way to ease the pain associated with sciatica.
The two primary types of surgery for sciatica are lumbar decompression and lumbar fusion. Lumbar decompression, which is more commonly performed, alleviates pressure on the sciatic nerve by taking out a section of the disc that is putting pressure on the nerve. Lumbar fusion is a more invasive surgery that involves fusing the vertebrae together to stabilize the spine and reduce pressure on the sciatic nerve.
After careful consideration and consultation with your physician, you may decide to have surgery for sciatica. Surgery should only be an option if less invasive methods have not alleviated pain and if there is proof of nerve damage.
Spinal Stenosis Diagnosis and Treatment
If you’re reading this, it’s likely that you or someone you know has been diagnosed with spinal stenosis. Although it’s not the most glamorous condition, don’t lose hope! There are treatment options available and, with the proper care, you can live a normal, healthy life.
So, let’s begin learning about spinal stenosis and how to treat it. We’ll go over everything from diagnosis to surgery to pain management.
Your healthcare provider may think that you have spinal stenosis based on your medical history and a physical exam. They may order one or more of the following imaging tests to confirm the diagnosis:
- An X-ray is a diagnostic test that uses electromagnetic energy beams to create images of internal tissues, bones, and organs onto film.
- Magnetic resonance imaging (MRI). A diagnostic procedure that uses a combination of large magnets, radio frequencies, and a computer to produce detailed images of organs and structures within the body.
- A computed tomography scan (CT or CAT scan) is a diagnostic imaging procedure that uses x-ray equipment to create detailed pictures, or slices, of the body. A CT scan provides more detail than an x-ray.
Imaging Tests
Your doctor may order one or more imaging tests to help diagnose spinal stenosis. These tests can help your doctor see if you have a narrowing of the spinal canal.
A CT scan is a medical imaging procedure that uses X-rays and computer technology to create detailed images of the inside of the body.
A CT scan employs X-rays and a computer to generate comprehensive pictures of your bones and soft tissues. A CT scan can reveal alterations in the spine that might be causing spinal stenosis, such as bone spurs or thickened ligaments.
Before the scan begins, you may be given contrast dye to help your spinal canal and other structures show up more clearly. The dye is injected into a vein in your arm.
Magnetic resonance imaging (MRI) is a powerful tool that can be used to diagnose a variety of conditions. MRI uses magnetic fields and radio waves to create detailed images of the inside of the body. MRI is safe and does not use ionizing radiation, making it a good choice for many patients.
An MRI uses a powerful magnet and radio waves to create detailed images of your spine. This type of scan can show whether you have stenosis and how severe it is. It can also reveal other issues with your spine, such as a herniated disk or tumors.
For this test, you may also receive contrast dye. MRI scanners are usually quite loud, so you may be given earplugs or headphones with music to listen to during the scan. If you’re claustrophobic (afraid of enclosed spaces), you may also be given a sedative.
A CT myelogram is a diagnostic test used to evaluate the spinal cord and surrounding structures. The test is performed by injecting contrast material into the spinal canal and then taking CT images. A CT myelogram is a type of CT scan that uses contrast dye injected into the space around your spinal cord. This provides your doctor with a clear view of your spinal canal and nerves.
Treatment
The answer to the question of how to treat spinal stenosis is not cut and dry. The treatment approach that is best for you will depend on several factors, such as the severity of your condition and your general health.
A combination of different treatments may be recommended by your doctor, such as:
- Physical therapy: A physical therapist can assist you in fortifying the muscles surrounding your spine and increasing your flexibility. You can also learn how to move without exacerbating your symptoms through physical therapy.
- Pain medication: If over-the-counter pain medications like ibuprofen or acetaminophen don’t work well, your doctor may prescribe stronger pain medications.
- Steroid injections: Injections of steroids can assist in decreasing inflammation and pain in the area affected.
- Surgery: In some cases, an operation may be the best way to take pressure off the spinal cord or nerves.
Medications
Medications may be prescribed by your healthcare provider to help alleviate the pain of spinal stenosis.
Some potential choices are:
- Antidepressants. Some people with moderate to severe pain may find relief with tricyclic antidepressants such as amitriptyline (Elavil, Endep) and nortriptyline (Pamelor).
- Opioids. These powerful painkillers include hydrocodone (Zohydro ER, Hysingla ER), oxycodone (OxyContin, Roxicodone), and morphine (MS Contin, Kadian). They’re generally only used if other medications haven’t been effective enough. Opioids can be addictive and cause other side effects like drowsiness, constipation, and nausea.
- Drugs for seizures. Medications such as gabapentin (Neurontin) and pregabalin (Lyrica), which were originally designed to treat seizure disorders, are often effective against the nerve damage that can cause chronic pain.
- Anti-inflammatory medications. These drugs – like celecoxib (Celebrex) and nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen (Advil, Motrin IB) and naproxen sodium (Aleve) – may provide some relief from the inflammation that’s causing your spinal stenosis pain.
- Milder forms of spinal stenosis pain may be relieved by over-the-counter medications such as acetaminophen (Tylenol, others).
Physical therapy
Spinal stenosis is a condition that can cause discomfort in the back and legs. Balance and flexibility can be improved with physical therapy, and a physical therapist can also show you exercises to help ease the pain.
Steroid Shots
Steroid shots are a common and effective treatment for spinal stenosis. Many people with this condition find that steroid shots help to relieve their pain and improve their quality of life. If you’re considering steroid shots for your own treatment, be sure to talk to your doctor about the potential risks and benefits.
Epidural steroid injections are most commonly administered by injecting the medication into the area around your nerve roots. This space is called the epidural space. The steroid medication helps reduce inflammation around your nerve roots and gives you more space in your spinal canal.
Injections of steroids may not be the best solution for everyone who has spinal stenosis. If you experience severe side effects from the injections or if the injections do not help to ease your pain, you might want to look into other treatment methods.
Surgery
Laminotomy – Your surgeon will take out a small sliver of bone from the lamina – the back section of one of your spinal bones – during this operation to create more room for your spinal cord. In certain situations, your surgeon might also put in metal fixtures to keep the bone steady.
Laminoplasty – This is a procedure that is similar to a laminotomy, but instead of your surgeon taking out a small wedge of bone, they will create an opening by cutting and folding back a section of the lamina. In the majority of cases, this operation is performed on both sides of the spine.
Laminectomy – Your surgeon will take out the whole lamina from one or more spinal bones during a laminectomy. This operation is commonly done in combination with a foraminotomy (explained below). Metal hardware may also be placed by your surgeon during this procedure to keep the remaining bones steady.
In some situations, your doctor may suggest a spinal fusion surgery. This involves joining two or more vertebrae together with metal hardware and a bone graft. The purpose of this surgery is to stabilize the spine and reduce pain.
Foraminotomy is a minimally invasive surgical procedure that involves making the foramen — the openings between the vertebrae where nerves pass through — larger. This surgery is often done along with other procedures, such as a laminectomy or spinal fusion.
The risks that come with any surgery, such as bleeding, infection, and blood clots, also apply to surgical procedures for spinal stenosis. In addition, each type of surgery for this condition comes with its own set of risks, which your surgeon will go over with you before the procedure.
Lifestyle and Home Remedies
Spinal stenosis can cause a great deal of pain and disability, but there are treatments available to ease your symptoms. Making changes to your lifestyle, taking medication and, in some instances, surgery can help you manage this condition.
Your doctor might first suggest lifestyle changes and home remedies help with your symptoms. For example, improving your posture, avoiding extended periods of sitting or standing, and losing weight can take pressure off your spine and relieve symptoms. Additionally, regular exercise helps keep your spine flexible.
In addition to making changes to your lifestyle, your doctor might also suggest taking over-the-counter pain relievers such as ibuprofen (Advil, Motrin IB) or naproxen sodium (Aleve). If these don’t provide relief, your doctor could prescribe a stronger medication.
If you have more severe symptoms, your doctor might suggest epidural steroid injections to help reduce inflammation around your spinal nerves. In some cases, surgery might be necessary to relieve pressure on your spinal cord or nerves. Your surgeon might recommend one of the following procedures:
Alternative Medicine
Spinal stenosis is often treated with medication and surgery by conventional means, but there are many alternative therapies that can be successful in managing the pain and discomfort associated with this condition. Integrative medicine takes a whole-body approach to spinal stenosis, using various modalities to provide relief.
Chiropractic care is often used to take pressure off the spine and enhance the range of motion. Massage therapy can help to loosen the muscles around the spine and ease the pain. acupuncture is another frequent treatment choice for those with spinal stenosis, as it can assist in releasing endorphins and reducing pain.
There are many other alternative therapies that can be effective in managing the pain of spinal stenosis. These include yoga, Tai Chi, and acupuncture. If you are considering any of these therapies, be sure to consult with your doctor first to ensure that they are safe for you to use.
What To Expect From Your Doctor
The initial step in diagnosis is frequently a meeting with your healthcare provider. They will probably inquire about your medical history and any symptoms you’re currently experiencing. They will also do a physical examination, which might involve tests to evaluate your range of motion, muscle strength, reflexes, and sensation.
Your doctor may also request imaging tests for a closer look at the structures in your spine. X-rays, MRIs, and CT scans are all possible options. These tests can help your doctor identify any narrowing of the spinal canal or other abnormalities.
After a diagnosis of spinal stenosis is made, you and your doctor can discuss treatment options. The severity of your symptoms and the cause of the condition will determine whether nonsurgical or surgical options are best.
Some nonsurgical treatments for spinal stenosis are:
- Activity modification: Adjust the activities you do to avoid worsening symptoms or causing pain.
- Weight loss: One way to reduce pressure on the spine is to lose any excess weight.
- Exercise: Exercises that don’t put a lot of strain on the spine and improve flexibility and strength.
- Medications: To reduce pain and inflammation, you may be prescribed pain relievers, anti-inflammatory drugs, and/or injections.
If your symptoms aren’t relieved by nonsurgical treatments or if your condition is causing neurological problems (like weakness, numbness, or loss of bowel or bladder control), surgery might be recommended. Surgery for spinal stenosis usually involves widening the spinal canal by taking out part of the vertebrae (decompression surgery) and/or stabilizing the spine with metal rods and screws (spinal fusion).
Herniated Disc Diagnosis and Treatment
You’ve arrived at the perfect destination if you’re in search of information on herniated disc diagnosis and treatment. Here, you’ll find all the details you need to know about this frequent condition, such as what it is, its causes, and available treatments.
Keep reading for everything you need to know about a herniated disc, whether you’re dealing with the condition yourself or are simply curious about it.
A herniated disc can usually be diagnosed by your doctor through a careful medical history and physical exam. In some cases, your doctor may order special tests like magnetic resonance imaging (MRI) or computed tomography (CT) scans.
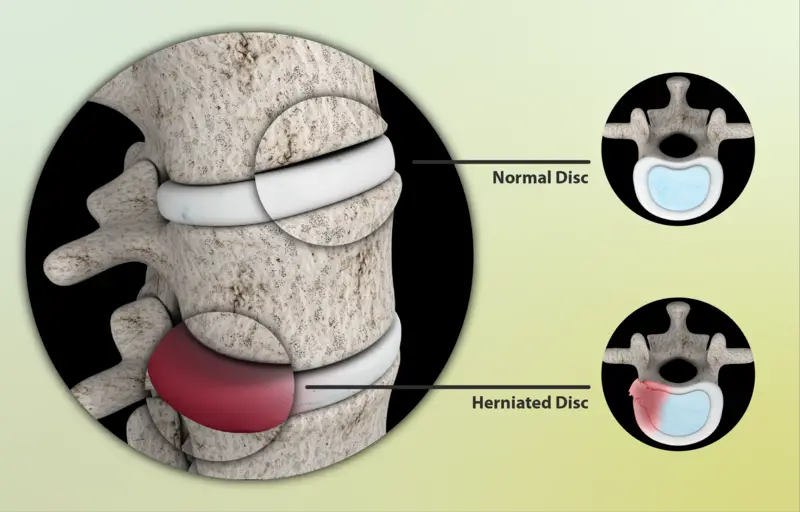
Your doctor will press on different areas of your spine to check for pain during the physical exam. They will also test your muscle strength and reflexes. To test muscle strength, you may be asked to lift your leg while lying down. To test reflexes, your doctor may tap your knee with a rubber hammer. These tests help show which nerves are being affected by the herniated disc.
The neurological exam is used to assess the function of your nervous system, including whether a herniated disc is pressing on a nerve. During this exam, you may be asked to walk on your toes or heels, stand on one leg, or raise your arms over your head. You may also be asked to turn your head from side to side or bend forward at the waist.
Imaging Tests
Imaging tests are frequently used to diagnose a herniated disc. These tests can give clear pictures of the spine and its internal structures.
A CT scan is a special x-ray that uses computer processing to create detailed images of your spine from different angles. Myelograms are often used along with CT scans.
An MRI uses a powerful magnetic field and radio waves to create images of your spine. MRI is very good at detecting herniated discs, but it is more expensive than a CT scan and is not available in all areas.
A myelogram is an x-ray of the spinal column after a dye has been injected into the area around the spinal cord. The dye makes it easier to see herniated discs or other issues with the spinal column on the x-ray. Myelograms are often combined with a CT scan.
Nerve Tests
Nerve tests are used to assess the electrical activity of nerves and muscles. Nerve conduction studies measure how well an electrical impulse passes through a nerve. An electromyogram (EMG) measures the electrical activity of muscles. The electrical activity of a muscle is measured by inserting a small needle electrode into it.
This can help diagnose:
- Nerve damage can be caused by a number of things, including diabetes, physical injury, and certain medications.
- Symptoms of nerve damage include numbness, tingling, and pain.
- Treatment for nerve damage depends on the underlying cause.
- Muscle tissue damage
- Nerves that are pinched
- Carpal tunnel syndrome
Guillain-Barré syndrome is a condition that can cause paralysis and other serious symptoms. If you think you may have this condition, it’s important to see a doctor right away. There is no cure for Guillain-Barré syndrome, but treatment can help improve your symptoms.
Treatment
The majority of herniated disc patients will try conservative treatment methods first. This typically includes a few days of rest, then pain medication and physical therapy. For the majority of people, this eliminates the symptoms and they can go back to living normally.
Medications
There are many medications that can be used to relieve the pain of a herniated disc. These include both over-the-counter and prescription drugs. If you are considering using medication to treat your herniated disc, talk to your doctor to see what options are available and right for you.
Short-term relief from mild to moderate pain can be achieved through the use of over-the-counter medications such as acetaminophen (Tylenol), ibuprofen (Advil, Motrin IB), and naproxen sodium (Aleve). However, it is important to only use them for short periods of time. Long-term use of these drugs can result in side effects like gastrointestinal bleeding and ulcers.
If you’re experiencing more severe pain, your doctor may prescribe opioids such as oxycodone (OxyContin, Percocet), hydrocodone (Vicodin), or morphine. Opioids are potent pain relievers, but they come with the risk of serious side effects like addiction and respiratory depression.
Spinal imaging should be done before beginning opioid therapy to make sure that the disc herniation is the root of your pain. If it is not, opioids could make your condition worse.
Other oral medications that may be prescribed for herniated disc pain include neuropathic drugs such as gabapentin (Neurontin) and pregabalin (Lyrica), muscle relaxants such as carisoprodol (Soma), and antidepressants such as duloxetine (Cymbalta) or venlafaxine (Effexor XR).
These drugs can help relieve pain by interfering with nerve impulses or relaxing muscles. They may also help improve sleep and reduce anxiety and depression, which can worsen the pain. Common side effects of these drugs include dizziness, drowsiness, dry mouth, nausea, and constipation.
Injecting cortisone into the afflicted area may also be recommended to diminish inflammation and pain. However, cortisone injections come with a risk of side effects, such as local skin reactions, infection, and nerve damage. Surgery might be required to take out the herniated disc or ease pressure on the spinal nerves in certain situations.
Therapy
If you have a herniated disc, your doctor may suggest physical therapy. Physical therapists are health care professionals who assess and treat patients with conditions that impede their movement and ability to do functional activities in their everyday lives.
A physical therapist will put together a treatment plan tailored to your individual needs. The objectives of physical therapy for a herniated disc are reducing pain and inflammation, encouraging healing, and helping you get back to normal function and range of motion. Treatment could involve exercises, stretches, traction, electrical stimulation, and hot or cold packs.
Surgery
If more conservative treatments haven’t worked and your herniated disc is causing severe pain or numbness, your doctor may suggest surgery. The entire disk must be removed in some cases. This is only done if the disk is severely damaged and unlikely to heal itself.
If the entire disk is removed, it can cause issues with spinal stability, so a bone graft to fuse the vertebrae together may be required. This can be done using your own bone or a donor’s bone. Metal hardware may also be employed to stabilize the spine.
If the inner portion of the disc isn’t harmed and there’s a decent chance it will heal by itself, only the part of the disc that’s sticking out is removed in other cases. You may have issues with bowel control or urinary incontinence after surgery, but these difficulties typically resolve themselves within a few weeks or months.
Although artificial disks are being investigated as a potential treatment for herniated discs, they are not yet widely available. It is unclear how effective they are, and there is a risk of loss of movement in the affected area.
Lifestyle and Home Remedies
excruciating, severe pain from a herniated disc can radiate down your leg and make even basic movements agonizing. You may be able to find some relief with these self-care measures:
Rest: When you first injure yourself, it’s important to avoid activities that put stress on your back. But after a few days, sitting or lying down for long periods of time can make stiffness and pain worse. Try mild activity such as a short walk.
For the first two days after an injury, apply cold packs to reduce swelling. After that, use heat to loosen up stiff muscles and joints.
Exercise: Exercise is not only beneficial for general fitness, but it can also help reduce back pain by reinforcing the muscles that support your back and increasing your flexibility and range of motion. Always warm up before you start working out and cool down afterward. Also, avoid any moves that result in pain. If an exercise makes your symptoms worse, stop immediately and consult your doctor or physical therapist.
If you’re experiencing short-term pain from a herniated disc, over-the-counter (OTC) medications like acetaminophen (Tylenol) or nonsteroidal anti-inflammatory drugs (NSAIDs) like ibuprofen (Advil, Motrin IB) can help. If OTC meds don’t do the trick, your doctor can prescribe stronger NSAIDs.
Alternative Medicine
Although there is little reliable evidence to support the use of alternative medicine treatments for herniated discs, some of these treatments may provide temporary relief.
Chiropractic care and spinal manipulation may provide moderate relief for certain types of neck pain and low back pain, according to a systematic review. The review found that chiropractic was usually modest in terms of short-term pain relief, with some trials showing no difference from sham treatments.
A systematic review concluded that massage may provide short-term relief from the pain associated with herniated discs. However, more research is needed to determine its long-term effectiveness.
Although acupuncture is sometimes used as a complementary medicine treatment for herniated discs, there is little evidence to support its efficacy. A systematic review found that acupuncture was no more effective than a placebo in the treatment of chronic neck pain.
What to Expect From Your Doctor
If you have symptoms of a herniated disc, it’s probably time to see your doctor. Your doctor will ask about your symptoms and how they began. Additionally, he or she may inquire about your bowel and bladder habits. A herniated disc in your lower back can sometimes interfere with bowel or bladder function, which is why this is the case.
If you use tobacco products, it’s important to be aware of the risks. Tobacco use can lead to a number of health problems, including cancer and heart disease. Quitting tobacco use is one of the best things you can do for your health. There are a number of resources available to help you quit, including counseling and medication.
If you’re thinking about quitting, talk to your doctor about the best way to do it. If you smoke cigarettes or use other tobacco products, your symptoms will become worse. Additionally, tobacco use can impede the healing process after surgery. As a result, your doctor will ask you to quit using tobacco before having any surgery to treat a herniated disc.
Your activity level and how often you lift heavy objects. Your doctor will ask about your level of activity and whether you have any occupations or hobbies that involve lifting heavy objects. Lifting heavy objects can aggravate a herniated disc.

 If you’re interested in
If you’re interested in